Dual channel interface chamber with inter-changeable templates for reduced dead-space

Description
This slice recording chamber is a dual version of the BSC2 designed to maintain isolated, living tissues in vitro in ‘interface’ mode and allow stable electrophysiological recordings to be made from the preparation. The temperature is controlled by a low noise proportional control heating unit, the PTC03 or in cases where only incubation or pharmacology is required, with a basic heating unit, the PTC04.
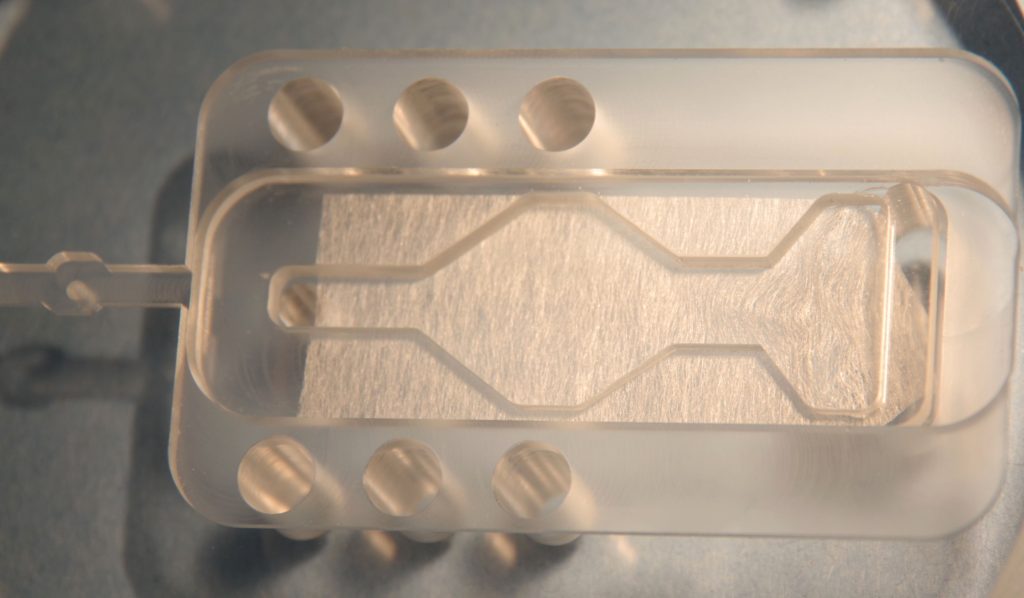
The chamber lower section is similar to the BSC2 for temperature control and oxygenation. Pre-oxygenated medium enters the main body of the chamber through a feed line, one to each well. This fine bore PTFE tube which spirals in the heated distilled water in the lower part of the chamber enters the upper part of the chamber. Here any bubbles in the line break and allow perfusion fluid to flow smoothly towards the slices. Acrylic templates allow the option for use of a single well or can be custom designed. The templates are positioned on the chamber acrylic base and optionally silicone grease is used to make the seal on the bottom (see diagram below). Typical dead space volume for the single well is approx. 200 ul. The length of PTFE tubing carrying the perfusion solution to the wells has a volume of approx. 400ul. The flow rate of the perfusion fluid is adjusted at the exit well by means of lens tissue which is used to wick solution out of the exit port. By adjusting the over-hang the flow rate is effectively adjusted and determines the characteristics of the meniscus of fluid around the slice preparation. As with all interface preparations, the high oxygen tension is maintained by bubbling a 95% oxygen, 5% carbon dioxide gas mixture through a ceramic bubbler located in the lower heated part of the chamber. This moistened and warmed gas mixture enters the upper part of the chamber via port-holes and is then deflected by an acrylic cover across to the centrally located slice preparation. The temperature in the upper chamber is maintained by ensuring that the medium and moistened gas mixture enter at the required temperature. This is dependent on the temperature of the lower chamber body which is warmed by a heating element controlled by the Proportional Temperature Controller, PTC03. An optional monitor sensor allows the upper chamber temperature to be checked when required. A more basic version of the controller PTC04 can be used instead where noise control is not an issue as with pre-incubation of slices in interface mode or for pharmacology experiments.
Features
- ‘Interface’ method of slice maintenance
- Two completely independent channels
- Use entire surface for multiple slices or reduced area with templates
- Single well diamond shaped templates, other designs available upon request
- Proportional temperature controller (PTC03) with low noise performance used for heating
- Optional basic temperature controller (PTC04) for interface slice incubation or pharmacology only
Schematic
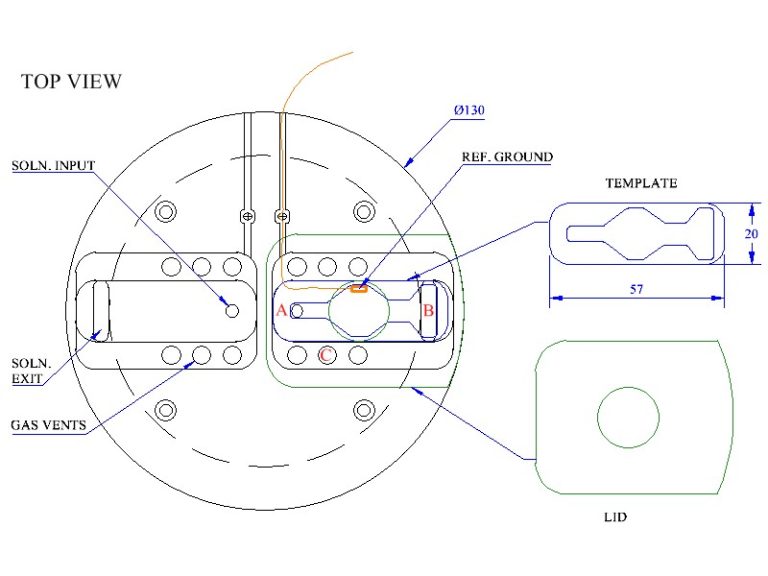
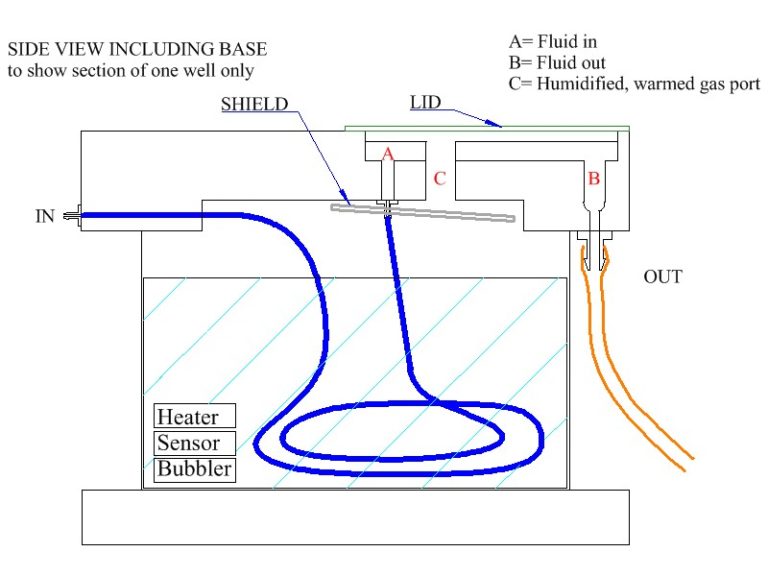
Typical arrangement of template and lens tissue in BSC2 chamber
Lens tissue is into a rectangle with a pointed end, the width should be equal to that of the template.


The lens tissue is placed into the base of the chamber well and the tip bent downwards into the exit well (below left). The lens tissue does not cover the solution inlet hole entirely so that any bubbles can escape to the atmosphere. Slices can rest on this lens tissue or optionally the template (below right) is placed over the lens tissue to restrict the flow more closely to the slice preparation. The chamber lid must be in place with the slices resting on the lens tissue in interface mode.